Facts on Flu Vaccines
Everyone 6 months and older needs a flu vaccine every year to protect yourself and your loved ones from getting sick with the flu.
It's not "just" the flu - it's a serious, but preventable, disease!

How effective is the flu vaccine?
Flu vaccines can reduce the chances of getting sick with the flu by up to 60%. Even if you still get sick, people who are vaccinated have less severe flu symptoms and are less likely to be hospitalized or die from flu-related complications.
Annual flu vaccination is the most important step to prevent getting and spreading the flu.
REDUCES
Illness from Flu
REDUCES
Flu Symptoms
REDUCES
Flu-Related Complications
How do we know that flu vaccines are safe?
Flu vaccines have been used since 1945 with very good safety records. Vaccines go through years of research and clinical trials before they are made available to the general public.

Can you get the flu from the flu vaccine?
Absolutely not. To cause infection, flu viruses need to reproduce, i.e., make copies of themselves. Flu vaccines, regardless of how they are made, do not contain viruses that can reproduce and make you sick with the flu.
What if I feel sick after getting a flu vaccine?
- You may be experiencing side effects of the vaccine, which should not be confused with actually being infected with the flu virus. A slight fever, aches, or fatigue can be a sign that your immune system is learning to fight off the flu. This is not flu. When someone is sick with flu they suffer severe and long-lasting symptoms.
- Since it can take up to two weeks following a flu vaccine for your body to build the proper immune response to help prevent flu, you could have been exposed to the flu virus before you were protected by the vaccine.
- You may be suffering from another infection that is not flu.

Parker's mom gets him a flu vaccine every year because flu took the life of the sister he will never know.
What are possible flu vaccine side effects?
Some people may experience redness and swelling at the site of injection, fever, headache, and/or muscle aches. These side effects are actually evidence that your body is responding to the vaccine to fight off a future flu infection. The risk of a severe allergic reaction in those who are vaccinated is less than one in one million!
Why do I need a new flu vaccine every year?
Because flu viruses are constantly changing, flu vaccines are updated from one season to the next to protect against the types of flu that are most likely to make people sick. Unlike other vaccines, flu vaccines are not intended to last longer than one year.
There are different types of vaccines such as:
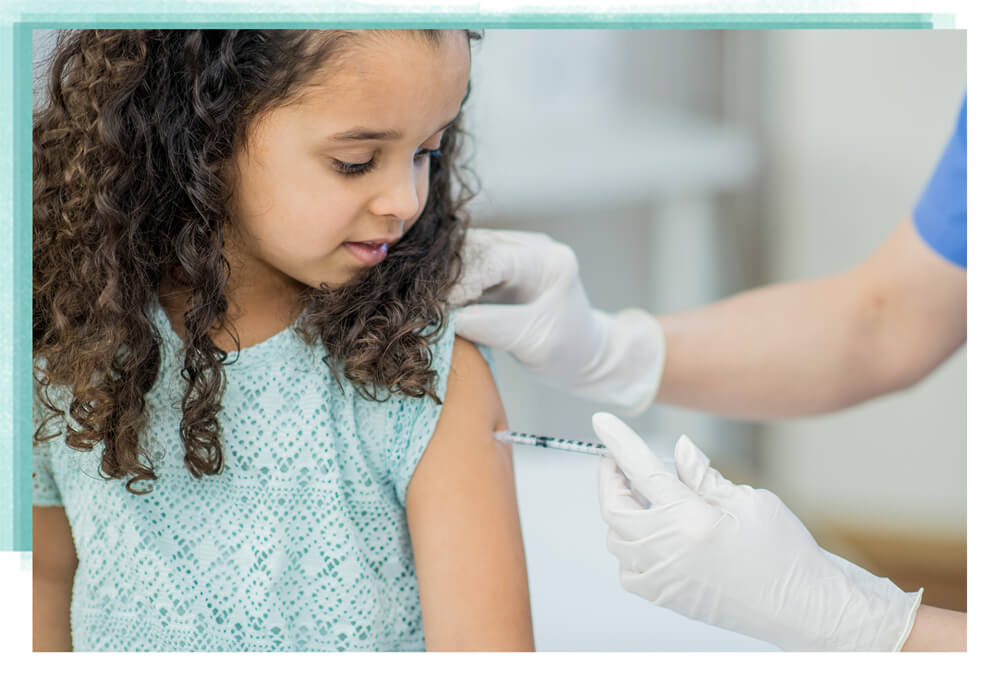
Inactivated vaccines contain a dead/killed virus that are administered as a “shot” with a needle.

Image source: CDC, Douglas Jordan, M.A., image provider.
Nasal spray vaccines contain a live, weakened flu virus that is sprayed into the nose instead of being administered as an injection.
Visit these other pages to learn more:
Page last reviewed: September 2021.


